Occurrence of acaricide resistance in Rhipicephalus microplus in brazilian municipalities: an integrative review
Abstract
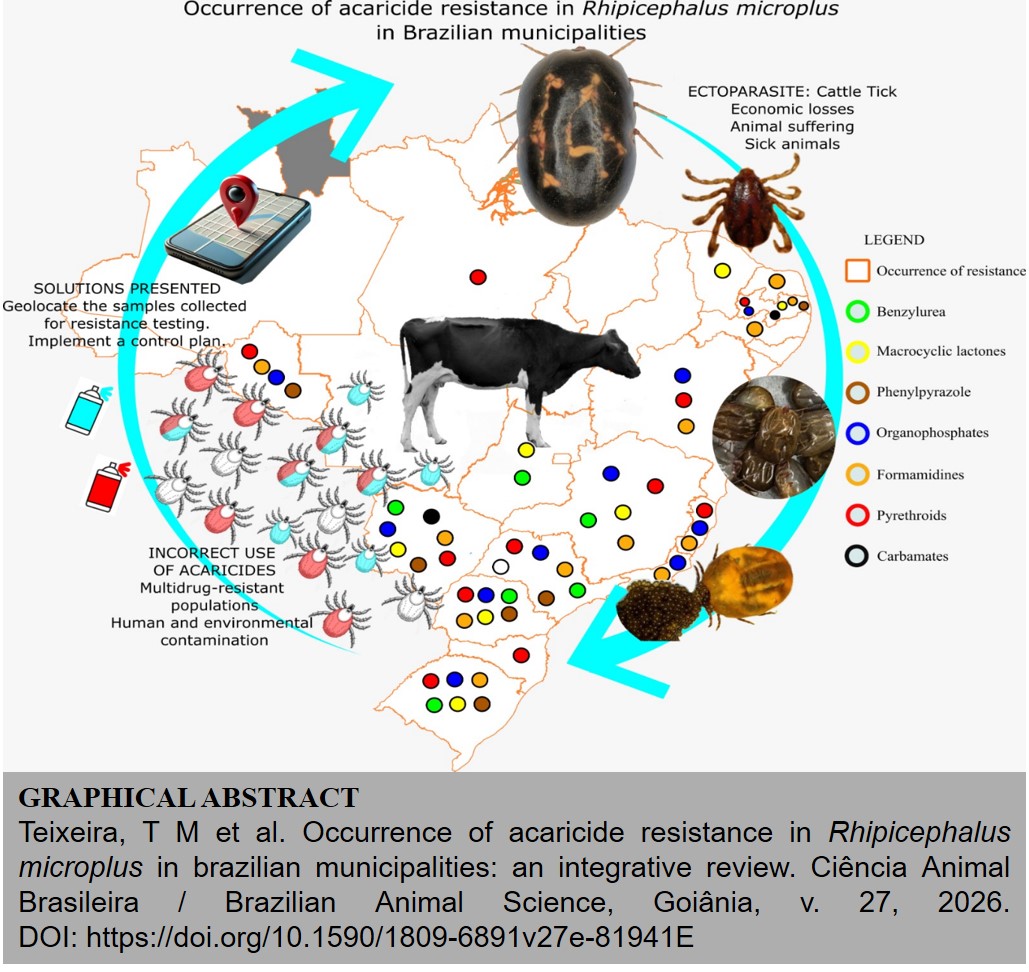
The objective of this study was to conduct an integrative review on the resistance of the tick Rhipicephalus microplus to synthetic acaricides, surveying the Municipalities where collections for resistance testing were carried out. The search was conducted in the Virtual Library of Veterinary Medicine and Animal Science (BVS-Vet), Google Scholar, CAPES Journal Portal, Web of Science, Scopus, and PubMed databases, considering publications between 2004 and 2025. Forty-six studies that met the inclusion criteria were selected. The analysis revealed that most of the research was conducted in the states of Rio Grande do Sul (69 Municipalities), Minas Gerais (66), and Mato Grosso do Sul (40). In the other states, few or no records were observed. The number of states, Municipalities, and publications found for pyrethroids were 11, 249, and 35; organophosphates 11, 145, and 28; carbamates 1, 37, 3; formamidine 12, 178, 27; phenylpyrazole 8, 65, 12; macrocyclic lactones 8, 27, 16; and benzylurea 6, 15, 10, respectively. Considering that livestock farming is present in 4,939 Brazilian Municipalities, available data on resistance are still limited. These results reinforce the need to implement public policies aimed at developing and implementing a national plan to combat cattle ticks.
Keywords: laboratory tests of acaricides; ectoparasites; geolocation; epidemiology.
Downloads
References
1. Miloca LM, Teixeira TM, Machado MA, Gazim ZC. Pesquisa de mercado de carrapaticidas comerciais para tratamento de bovinos nos municípios de Perobal, Umuarama e Xambrê – Paraná, Brasil. Informe GEPEC. 2024;28(2):516-36. doi: https://doi.org/10.48075/igepec.v24i2
2. Grisi L, Leite RC, Martins JR, Barros ATM, Andreotti R, Cançado PHD, et al. Reassessment of the potential economic impact of cattle parasites in Brazil. Rev Bras Parasitol Vet. 2014;23(2):150-6. doi: https://doi.org/10.1590/S1984-29612014042
3. Calvano MPCA, Brumatti RC, Barros JC, Garcia MV, Martins KR, Andreotti R. Bioeconomic simulation of Rhipicephalus microplus infestation in different beef cattle production systems in the Brazilian Cerrado. Agric Syst. 2021;194:103247. doi: https://doi.org/10.1016/j.agsy.2021.103247
4. Singh K, Kumar S, Sharma AK, Jacob SS, Verma MR, Singh NK, et al. Economic impact of predominant ticks and tick-borne diseases on Indian dairy production systems. Exp Parasitol. 2022;243:108408. doi: https://doi.org/10.1016/j.exppara.2022.108408
5. Morais PGS, Silva MCA, Gonçalves MF, Ambrósio A, Fagundes NS, Nascimento MRBM. A influência da coevolução bovinos/carrapatos nos métodos de controle e ambiência na bovinocultura. PUBVET. 2013;7(9):1534. doi: https://doi.org/10.22256/pubvet.v7n9.1534
6. Instituto Brasileiro de Geografia e Estatística. Bovinos (bois e vacas): tamanho do rebanho – 2023. Brasília: IBGE; 2023. Disponível em: https://www.ibge.gov.br/explica/producao-agropecuaria/bovinos/br
7. Formigoni I. Preço do boi gordo, bezerro, milho e soja: média anual entre 2018 e 2024. FarmNews; 2025 [citado 2025 maio 2]. Disponível em: https://www.farmnews.com.br
8. Vanderstichel R, Dohoo I, Sanchez J, Sithole F, Keefe G, Stryhn H. Predicting the effect of anthelmintic treatment on milk production of dairy cattle in Canada using an Ostertagia ostertagi ELISA. Prev Vet Med. 2013;111(1-2):63-75. doi: https://doi.org/10.1016/j.prevetmed.2013.04.001
9. Jonsson NN. The productivity effects of cattle tick (Boophilus microplus) infestation on cattle. Vet Parasitol. 2006;137(1-2):1-10. doi: https://doi.org/10.1016/j.vetpar.2006.01.010
10. Jongejan F, Uilenberg G. The global importance of ticks. Parasitology. 2004;129(Suppl 1):S3-14. doi: https://doi.org/10.1017/S0031182004005967
11. Brito LG, Silva Neto FG, Oliveira MCS, Barbieri FS. Bioecologia, importância médico-veterinária e controle de carrapatos, com ênfase no carrapato dos bovinos Rhipicephalus (Boophilus) microplus. Porto Velho: Embrapa Rondônia; 2006. 24 p.
12. Brasil. Ministério da Agricultura, Pecuária e Abastecimento. Instrução Normativa nº 56, de 6 de novembro de 2008. Diário Oficial da União; 2008. Disponível em: https://www.gov.br/agricultura
13. Dedavid e Silva LA. Vaccination against Rhipicephalus microplus: an alternative to chemical control? Cienc Rural. 2024;54(3):e20230161. doi: https://doi.org/10.1590/0103-8478cr20230161
14. Andreotti R, Garcia MV, Koller WW. Carrapatos na cadeia produtiva de bovinos. Brasília: Embrapa; 2019.
15. Rodriguez-Vivas RI, Trees AJ, Rosado-Aguilar JA, Villegas-Perez SL, Hodgkinson JE. Evolution of acaricide resistance in field populations of Rhipicephalus microplus. Int J Parasitol. 2011;41(8):895-903. doi: https://doi.org/10.1016/j.ijpara.2011.03.012
16. Bardosh K, Waiswa C, Welburn S. Conflict of interest: use of pyrethroids and amidines against tsetse and ticks in zoonotic sleeping sickness endemic areas of Uganda. Parasites Vectors. 2013;6:204. doi: https://doi.org/10.1186/1756-3305-6-204
17. Koller WW. Resistência dos carrapatos aos acaricidas. In: Andreotti R, Garcia MV, Koller WW, editores. Carrapatos na cadeia produtiva de bovinos. Brasília: Embrapa; 2019. p. 149-60.
18. Abbas RZ, Zaman MA, Colwell DD, Gilleard J, Iqbal Z. Acaricide resistance in cattle ticks and approaches to its management. Vet Parasitol. 2014;203(1-2):6-20. doi: https://doi.org/10.1016/j.vetpar.2014.03.006
19. Medeiros JP, Bortollucci WC, Silva ES, Oliveira HLM, Gonçalves JE, Junior RP, et al. Biocidal potential of Eugenia pyriformis essential oil against Rhipicephalus microplus. Pesq Vet Bras. 2019;39(11):879-88. doi: https://doi.org/10.1590/1678-5150-PVB-6434
20. Quadros DG, Johnson TL, Whitney TR, Oliver JD, Chávez ASO. Plant-derived natural compounds for tick pest control. Insects. 2020;11(8):490. doi: https://doi.org/10.3390/insects11080490
21. Santos FCC, Vogel FSF, Roll VFB, Monteiro SG. In vitro effect of plant extracts on Rhipicephalus microplus. Cienc Anim Bras. 2013;14(1):113-9. doi: https://doi.org/10.5216/cab.v14i1.19393
22. Koul O. Neem: a global perspective. In: Koul O, Wahab S, editors. Neem: today and in the new millennium. Dordrecht: Springer; 2004. p. 1-19. doi: https://doi.org/10.1007/1-4020-2596-3_1
23. Andreotti R, Gomes A, Malavazi-Piza KC, Sasaki SD, Sampaio CAM, Tanaka AS. BmTI antigens induce a bovine protective immune response. Int Immunopharmacol. 2002;2(4):557-63. doi: https://doi.org/10.1016/S1567-5769(01)00203-X
24. Russell CL. An overview of the integrative research review. Prog Transplant. 2005;15(1):8-13. doi: https://doi.org/10.1177/152692480501500102
25. World Health Organization. Resistance of malaria parasites to drugs. WHO Tech Rep Ser. 1965;296:29.
26. Raynal JT, Borges AA, Bastos BL, Bahiense TC, Meyer R, Portela RW. Avaliação da eficiência de acaricidas na Bahia. PUBVET. 2018;12:1-8. doi: https://doi.org/10.31533/pubvet.v12n9a163
27. Santana BB, Ramos RAN, Faustino MAG, Alves LC, Carvalho GA. Evaluation of cypermethrin and amitraz efficacy. Arq Inst Biol. 2015;82:1-4. doi: https://doi.org/10.1590/1808-1657001202013
28. Raynal JT, Bastos BL, Silva MC, Bahiense TC, Meyer R, Portela RW. Acaricide efficacy over five years. PUBVET. 2020;14(4):1-3. doi: https://doi.org/10.31533/pubvet.v14n4a546
29. Higa LOS, Garcia MV, Barros JC, Koller WW, Andreotti R. Resistance of Rhipicephalus microplus to acaricides. Rev Bras Parasitol Vet. 2016;25(2):163-71. doi: https://doi.org/10.1590/S1984-29612016026
30. Marchesini P, Novato TP, Cardoso SJ, Prata MCA, Nascimento RM, Klafke G, et al. Acaricidal activity of cinnamaldehyde and α-bisabolol. Vet Parasitol. 2020;286:109226. doi: https://doi.org/10.1016/j.vetpar.2020.109226
31. Carneiro JC, Costa EGL, Vasconcelos VO, Oliveira NJF, Duarte ER. Diagnóstico do controle de acaricidas. Acta Sci Vet. 2015;43:1-10
32. Zaldivar MF, Bastianetto E, Pereira Filho AA, Rodrigues DS, Martins Júnior VS, Morais-Costa F, et al. Acaricide effect of plants from the Brazilian savanna. Vet Parasitol. 2024;331:110281. doi: https://doi.org/10.1016/j.vetpar.2024.110281
33. Machado FA, Pivoto FL, Ferreira MST, Gregorio FV, Vogel FSF, Sangioni LA. Multiresistant Rhipicephalus microplus. Rev Bras Parasitol Vet. 2014;23:337-42. doi: https://doi.org/10.1590/S1984-29612014063
34. Domingues LN, Brasil BSAF, Bello ACPP, Cunha AP, Barros TM, Leite RC, et al. Survey of resistance in Brazilian ticks. Vet Parasitol. 2012;189:327-32. doi: https://doi.org/10.1016/j.vetpar.2012.04.031
35. Mastrantonio EC, Pereira DA, Caixeta PCMM, Maia MB. Eficiência de acaricidas comerciais. Vet Not. 2022:1-10.
36. Junior PB, Barros JC, Maciel WG, Garcia MV, Higa LOS, Andreotti R. Control strategies and economic evaluation. Acta Parasitol. 2022;67(4):1564-72. doi: https://doi.org/10.1007/s11686-022-00611-8
37. Gomes A, Koller WW, Barros ATM. Suscetibilidade a carrapaticidas. Cienc Rural. 2011;41:1447-52. doi: https://doi.org/10.1590/S0103-84782011005000105
38. Valsoni LM, Freitas MG, Echeverria JT, Borges DGL, Tutija J, Borges FA. Resistance to all chemical groups. Int J Acarol. 2020;46(4):276-80. doi: https://doi.org/10.1080/01647954.2020.1765867
39. Sousa ABB, Bianchi D, Santos EM, Dias SR, Peleja PL, Santos RR, et al. First description of acaricide resistance in the lower Amazon. Animals. 2022;12(21):2931. doi: https://doi.org/10.3390/ani12212931
40. Silva R, Santos MPA, Figueiredo MAP. Avaliação comparativa in vitro da sensibilidade a acaricidas. Ars Vet. 2020;36(3):163-8. doi: https://doi.org/10.15361/2175-0106.2020v36n3p163-168
41. Vilela VLR, Feitosa TF, Bezerra RA, Klafke GM, Riet-Correa F. Multiple acaricide-resistant ticks. Ticks Tick Borne Dis. 2020;11(4):101413. doi: https://doi.org/10.1016/j.ttbdis.2020.101413
42. Silva EHA, Brito RS, Santos AJ, Silva Junior EB, Siqueira IVM, Xavier CMO, et al. Performance of synthetic acaricides and essential oil. Arq Bras Med Vet Zootec. 2024;76:1-6. doi: https://doi.org/10.1590/1678-4162-13160
43. Santana LF, Carvalho Júnior RA, Moreira ÉFA, Rodrigues EA, Parreira Filho JM, Freitas BBB, et al. Regional study of acaricides. Innov Sci Technol J. 2019:51-5.
44. Dolenga CJR, Anjos A, Barbosa VHG, Yoshitani UY, Castilho PLSP, Miyakawa VI, et al. Acaricidal effect of major compounds. Rev Bras Parasitol Vet. 2022;31(2):e006022. doi: https://doi.org/10.1590/S1984-29612022028
45. Brito LG, Barbieri FS, Rocha RB, Oliveira MC, Ribeiro ES. Evaluation of acaricide efficacy in dairy herds. Vet Med Int. 2011;2011:806093. doi: https://doi.org/10.4061/2011/806093
46. Gasparotto PHG, Santos CAF, Dantas Filho JV, Ferraz RCS, Silva FRC, Daudt C. Resistance of Rhipicephalus microplus in Rondônia. Acta Vet Bras. 2020;14(2). doi: https://doi.org/10.21708/avb.2020.14.2.9123
47. Duarte FC, Roma Junior LC, Santos ML, Fiorini LC, Mendes MC. Influence of extension activities on tick control. Arq Inst Biol. 2021;88:1-9. doi: https://doi.org/10.1590/1808-1657000482020
48. Pedrassani D, Reisdorfer S. Avaliação da eficácia in vitro de carrapaticidas. Arch Vet Sci. 2015;20:17-29. doi: https://doi.org/10.5380/avs.v20i2.37195
49. Reck J, Klafke GM, Webster A, Dall’Agnol B, Scheffer R, Souza UA, et al. First report of fluazuron resistance. Vet Parasitol. 2014;201(1-2):128-36. doi https://doi.org/:10.1016/j.vetpar.2014.01.012
50. Klafke G, Webster A, Dall’Agnol B, Pradel E, Silva J, La Canal LH, et al. Multiple resistance to acaricides. Ticks Tick Borne Dis. 2017;8(1):73-80. doi: https://doi.org/10.1016/j.ttbdis.2016.09.019
51. Camillo G, Vogel FSF, Sangioni LA, Cadore GC, Ferrari R. Eficiência in vitro de acaricidas. Cienc Rural. 2009;39:490-5. doi: https://doi.org/10.1590/S0103-84782008005000082
52. Jongejan F, Berger L, Reck J, Ferreira PT, Jesus MS, Scott FB, et al. RaTexT®: a rapid tick exposure test. Parasites Vectors. 2024;17:365. doi: https://doi.org/10.1186/s13071-024-06448-6
53. Ueno TEH, Mendes EEB, Pomaro SHK, Lima CKP, Guilloux AGA, Mendes MC. Sensitivity profile of ticks to acaricides. Arq Inst Biol. 2012;79:177-83
54. Torres-Santos PT, Farias IF, Passos GS, Almeida MD, Horta MC. Avaliação in vitro da resistência. Vet Zootec. 2021;28:1-9. doi: https://doi.org/10.35172/rvz.2021.v28.550
55. Moraes N, Nicaretta JE, Rodrigues DC, Gonzaga BCF, Barrozo MM, Vale FL, et al. Comparison of acaricide application methods. Ticks Tick Borne Dis. 2023;14(4):102190. doi: https://doi.org/10.1016/j.ttbdis.2023.102190
56. Dolenga CJR, Anjos A, Yoshitani UY, Molento MB. Análise in vitro da resistência a acaricidas. In: Melchior LAK, Silva Malavazi PFN, Camargo LMA, et al., editores. Atualidades em medicina tropical no Brasil. Rio Branco: Stricto Sensu; 2020. p. 65-79.
57. Daher DO, Bertolucci AV, Lopes E, Guimarães AM, Rocha CMBM. Fatores associados à resistência. Rev Verde Agroecol Desenvolv Sustent. 2012;7(1):102-15.
58. Coelho WAC, Pereira JS, Fonseca ZAAS, Andre WPP, Bessa EN, Paiva KAR, et al. Resistência à cipermetrina e amitraz. Acta Vet Bras. 2013;7(3):229-32.
59. Mendes EC, Mendes MC, Sato ME. Diagnosis of amitraz resistance. Exp Appl Acarol. 2013;61:357-69. doi: https://doi.org/10.1007/s10493-013-9697-3
60. Maciel WG, Lopes WDZ, Cruz BC, Gomes LV, Teixeira WF, Buzzulini C, et al. Effectiveness of amitraz after ten years. Vet Parasitol. 2015;214(3-4):233-41. https://doi.org/doi:10.1016/j.vetpar.2015.10.024
61. Salvador VF, Morais IML, Leal LLLL, Tamiozo GL, Chagas HDF, Silva IS, et al. Resistance of Rhipicephalus microplus in tropical climates. Vet Parasitol. 2025;336:110441. doi: https://doi.org/10.1016/j.vetpar.2025.110441
62. Lopes WDZ, Chiummo RM, Vettorato LF, Rodrigues DC, Sonada RB. Effectiveness of fipronil + fluazuron. Parasitol Int. 2017;66(5):627-34. doi: https://doi.org/10.1016/j.parint.2017.05.005
63. Gomes LVC, Lopes WDZ, Cruz BC, Teixeira WF, Felippelli G, Maciel WG, et al. Acaricidal effects of fluazuron and ivermectin. Exp Parasitol. 2015;153:22-8. doi: https://doi.org/10.1016/j.exppara.2015.02.004
64. Ferreira LC, Lima EF, Silva ALP, Feitosa TF, Klafke GM, Vilela VLR. Effect of cyclosporin A on toxicity of macrocyclic lactones. Ticks Tick Borne Dis. 2023;14(2):102113. doi: https://doi.org/10.1016/j.ttbdis.2022.102113
65. Ferreira LC, Lima EF, Silva ALP, Oliveira CSM, Filho GMS, Sousa LC, et al. Cross-resistance between macrocyclic lactones. Exp Appl Acarol. 2022;87(1):109-17. doi: https://doi.org/10.1007/s10493-022-00730-x
66. Maciel WG, Lopes WDZ, Gomes LVC, Cruz BC, Felippelli G, Santos IBD, et al. Susceptibility to fluazuron and novaluron + eprinomectin. Prev Vet Med. 2016;135:74-86. doi: https://doi.org/10.1016/j.prevetmed.2016.10.019
67. Cruz BC, Lopes WDZ, Maciel WG, Felippelli G, Fávero FC, Teixeira WFP, et al. Susceptibility to ivermectin. Vet Parasitol. 2015;207(3-4):309-17. doi: https://doi.org/10.1016/j.vetpar.2014.12.012
68. Vargas MS, Céspedes NS, Sánchez HF, Martins JR, Céspedes COC. Avaliação in vitro de cepa resistente ao amitraz. Cienc Rural. 2003;33(4):737-42. doi: https://doi.org/10.1590/S0103-84782003000400024
69. Cruz BC, Gomes LVC, Maciel WG, Felippelli G, Santos IBD, Cruvinel LB, et al. In vivo effect of diflubenzuron. Rev Bras Parasitol Vet. 2018;27:545-54. doi: https://doi.org/10.1590/S1984-296120180084
70. Instituto Brasileiro de Geografia e Estatística. Pesquisa da Pecuária Municipal: tabelas 2022. Brasília: IBGE; 2022. Disponível em: https://www.ibge.gov.br
71. Skieresinski J. Secretaria da Agricultura lança vídeo sobre controle de carrapatos. Porto Alegre: SEAPI; 2018 [citado 2025 jun 25]. Disponível em: https://www.agricultura.rs.gov.br
72. Universidade Federal de Santa Maria. Programa de Pós-Graduação em Medicina Veterinária. Santa Maria: UFSM; 2025 [citado 2025 jun 25]. Disponível em: https://www.ufsm.br
73. Instituto de Pesquisas Veterinárias Desidério Finamor. Pesquisadores. Eldorado do Sul: IPVDF; 2025 [citado 2025 jun 25]. Disponível em: http://www.ipvdf.rs.gov.br
74. Gomes CCG. O carrapato-do-boi e o manejo da resistência aos carrapaticidas. Comunicado Técnico nº 70. Bagé: Embrapa Pecuária Sul; 2009.
75. Brito LG. Como realizar a coleta e o envio de carrapatos para o teste de eficácia. Bagé: Embrapa; 2006.
76. Instituto Brasileiro de Geografia e Estatística. Produção de leite: valor da produção – 2023. Brasília: IBGE; 2023. Disponível em: https://www.ibge.gov.br
77. Gomes CCG. Instruções para coleta e envio de material para teste de sensibilidade. Bagé: Embrapa Pecuária Sul; 2010.
78. Valotto AA. Melhoramento genético em bovinocultura de leite. Curitiba: SENAR-PR; 2018. 108 p.
79. Sobral A, Souza-Santos R. Importância do geoprocessamento nas análises em saúde. Salvador: UFBA; 2021. Disponível em: https://nehma.ufba.br
80. Santana BB, Ramos RAN, Santana MA, Alves LC, Carvalho GA. Susceptibility of Rhipicephalus microplus to pyrethroids. Rev Bras Parasitol Vet. 2013;22(2):1-5. doi: https://doi.org/10.1590/S1984-29612013000200047
81. Souza MI, Soares GSL, Conceição AI, Silva JCR, Dantas AFM, Cajueiro JFP, et al. Diseases with neurological manifestations in cattle. Cienc Anim Bras. 2023;24(1):e76190. doi: https://doi.org/10.1590/1809-6891v24e-76190E
82. Furlong J, editor. Carrapato: problemas e soluções. Juiz de Fora: Embrapa Gado de Leite; 2005. 65 p.
83. Lorenz C, Virginio F, Lima GC, organizadores. Viver no futuro: ainda tem clima para isso? São Paulo: Ed. dos Autores; 2021.
84. Siqueira F, Blecha IMZ, Cardoso FF. Variabilidade genética da resistência bovina ao carrapato. In: Andreotti R, Garcia MV, Koller WW, editores. Carrapatos na cadeia produtiva de bovinos. Brasília: Embrapa; 2019. p. 225-40.
85. Van Dalen EMSP, Jansen Van Rensburg C. Competitive displacement and acaricide resistance. Exp Appl Acarol. 2024;92:135-49. doi: https://doi.org/10.1007/s10493-023-00871-7
86. Porta M, editor. A dictionary of epidemiology. 6th ed. Oxford: Oxford University Press; 2014. 343 p.
87. Santos TR, Klafke GM, Pappen FG, Nizoli LQ, Biegelmeyer P, Farias NAR. Comparison of larval bioassays. Rev Bras Parasitol Vet. 2013;22(4):495-501. doi: https://doi.org/10.1590/S1984-29612013000400008
Bras Parasitol Vet. 2013;22(4):495-501. doi: https://doi.org/10.1590/S1984-29612013000400008
88. Instituto Nacional de Pesquisas Espaciais. SPRING: sistema de processamento de informações georreferenciadas. São José dos Campos: INPE; 2023 [citado 2025 out 2]. Disponível em: http://www.dpi.inpe.br
89. Anselin L, Syabri I, Kho Y. GeoDa: an introduction to spatial data science. 2025 [citado 2025 out 2]. Disponível em: https://geodacenter.github.io
90. Wiens J, Shenoy ES. Machine learning for healthcare epidemiology. Clin Infect Dis. 2018;66(1):149-53.
Downloads
Additional Files
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Brazilian Animal Science/ Ciência Animal Brasileira

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors who publish with this journal agree to the following terms:
- Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
- Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgement of its initial publication in this journal.
- Authors are permitted and encouraged to post their work online (e.g. in institutional repositories or on their website) prior to and during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work (See The Effect of Open Access).
Data statement
-
The research data is available on demand, condition justified in the manuscript



























