Resistência a antimicrobianos e tolerância ao arsênio em Enterococcus spp. isolados de Leopardus geoffroyi de vida livre em Candiota, no bioma Pampa, Brasil: sentinelas da poluição ambiental
Resumo
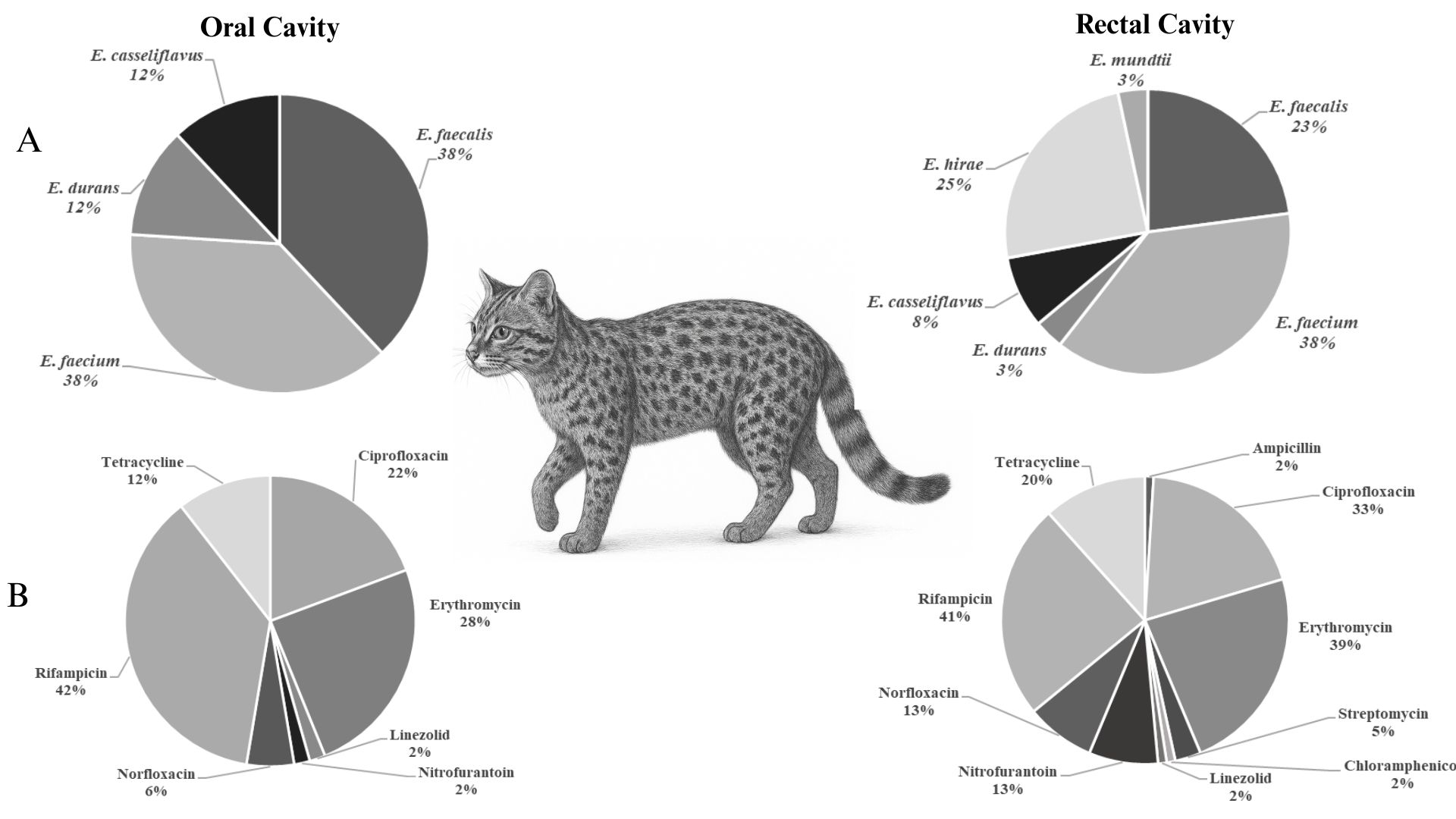
O bioma Pampa abriga uma notável diversidade de espécies endêmicas, mas vem sofrendo intensos impactos decorrentes de atividades antrópicas, que ameaçam a conservação de espécies silvestres, tais como o gato-do-mato-grande (Leopardus geoffroyi). Nesse contexto, este estudo avaliou a resistência a antimicrobianos e a tolerância a metais pesados em enterococos isolados das cavidades oral e retal de 14 gatos-do-mato-grande (L. geoffroyi) de vida livre da região do Seival/Candiota, no bioma Pampa, como bioindicadores da qualidade ambiental. O isolamento foi realizado em meios seletivos, a identificação das espécies foi feita por MALDI-TOF, e a suscetibilidade foi determinada pelo método de disco-difusão frente a 12 antimicrobianos de uso clínico e veterinário. Genes de resistência a antimicrobianos (msrC, ermB, tetM e tetL) e de tolerância a metais pesados (arsA_I, arsA_II e tcrB) foram investigados por PCR. Um total de 111 isolados de enterococos foi recuperado. Enterococcus faecium (37,8 %) e Enterococcus faecalis (29,7 %) predominaram, apresentando distribuição distinta entre amostras orais e retais. No geral, 74,8 % dos isolados apresentaram resistência a pelo menos um antimicrobiano, principalmente à rifampicina (41,4 %), à eritromicina (34,2 %) e à ciprofloxacina (26,1 %). Não foi detectada resistência à gentamicina nem à vancomicina. A resistência a múltiplos antimicrobianos foi observada em 17 isolados (20,5 %). Determinantes de resistência (msrC, ermB, tetM e tetL) e o gene de tolerância ao arsênio (arsA_I) foram identificados, enquanto arsA_II e tcrB não foram detectados. Em conclusão, a presença de enterococos resistentes a múltiplos antimicrobianos e tolerantes ao arsênio em gatos-do-mato-grande de vida livre reforça seu uso como bioindicadores da qualidade ambiental e evidencia o impacto das atividades antrópicas na saúde animal.
Palavras-chave: Impactos antropogênicos; bioindicadores ambientais; conservação da vida silvestre; saúde única.
Downloads
Referências
1. Bilenca D, Miñarro F. Identificación de Áreas Valiosas de Pastizal (AVPs) en las Pampas y campos de Argentina, Uruguay y sur de Brasil. Argentina; 2004. Available from: http://awsassets.wwfar.panda.org/downloads/libro_avps__bilenca_y_minarro_2004_.pdf
2. Instituto Brasileiro de Geografia e Estatística (IBGE). Biomas e sistema costeiro-marinho do Brasil. Rio de Janeiro: IBGE; 2019. Available from: https://biblioteca.ibge.gov.br/visualizacao/livros/liv101676.pdf
3. Andrade BO, Dröse W, Aguiar CAD, et al. 12,500+ and counting: biodiversity of the Brazilian Pampa. Front Biogeogr 2023 15.2, e59288. Available from: https://doi.org/10.21425/F5FBG59288
4. Centro De Sensoriamento Remoto (CRS), Instituto Nacional De Pesquisas Espaciais (INPE). Relatório técnico do monitoramento do desmatamento do Bioma Pampa 2008-2009. Brasília: Ministério do Meio Ambiente (MMA); 2010. Available from: https://antigo.mma.gov.br/estruturas/sbf_chm_rbbio/_arquivos/relatrio_tcnico_monitoramento_pampa_2008_2009_72.pdf
5. Ziliotto M, Kulmann-Leal B, Roitman A, et al. Pesticide pollution in the Brazilian Pampa: Detrimental impacts on ecosystems and human health in a neglected biome. Pollutants 2023;3(2):280–292. Available from: https://doi.org/10.3390/pollutants3020020
6. Stockwell S. Working in Brazilian wildcat conservation with Flavia Tirelli 2023; Available from: Available from: https://www.conservation-careers.com/interviews/working-in-brazilian-wildcat-conservation-with-flavia-tirelli/
7. International Union For Conservation Of Nature (IUCN). The IUCN Red List of Threatened Species. Version 2025-1. 2025. Available from: https://www.iucnredlist.org/
8. Brasil. Ministério do Meio Ambiente. Portaria MMA no. 148, de 7 de junho de 2022. Lista nacional oficial de espécies da fauna ameaçadas de extinção. Brasília, DF: Diário Oficial da União, 2022. Available from: https://in.gov.br/en/web/dou/-/portaria-mma-n-148-de-7-de-junho-de-2022-406272733
9. Governador Do Estado Do Rio Grande Do Sul. Decreto no. 51.797, de 8 de setembro de 2014. Espécies da fauna silvestre ameaçadas de extinção do Rio Grande do Sul. Porto Alegre: Governo do Estado do Rio Grande do Sul, 2014. Diário Oficial do Estado do Rio Grande do Sul DOE nº 173, de 09 de setembro de 2014. Available from: https://leisestaduais.com.br/rs/decreto-n-51797-2014-rio-grande-do-sul-declara-as-especies-da-fauna-silvestre-ameacadas-de-extincao-no-estado-do-rio-grande-do-sul. Acesso em: 18 fev. 2026
10. Pereira J, Lucherini M, Trigo T. Leopardus geoffroyi. The IUCN Red List of Threatened Species [Homepage on the Internet]. IUCN Red List Threat. Species. 2015; Available from: https://www.iucnredlist.org/species/15310/50657011
11. Peters FB, Mazim F, Favarini, MO, et al. Caça preventiva ou retaliativa de felinos por humanos no extremo sul do Brasil. In: Castaño-Uribe C, editor. Conflictos entre felinos y humanos en América Latina. Bogotá: Instituto de Investigación de Recursos Biológicos Alexander von Humboldt (IAvH), 2016; p. 311–325.
12. Tirelli FP, Mazim FD, Crawshaw Jr PG, et al. Density and spatio-temporal behaviour of Geoffroy’s cats in a human-dominated landscape of southern Brazil. Mamm Biol 2019;99(1):128–135. Available from: https://doi.org/10.1016/j.mambio.2019.11.003
13. Silver S. Bacterial resistances to toxic metal ions: a review. Gene 1996;179(1):9–19. Available from: https://doi.org/10.1016/S0378-1119(96)00323-X
14. Ienes-Lima J, Prichula J, Abadie M, et al. Anthropic impact on the critically endangered Melanophryniscus admirabilis (Admirable Redbelly Toad): evidence from the presence of multiresistant enterobacteriaceae. Curr Microbiol. 2023;80(11):339. Available from: https://doi.org/10.1007/s00284-023-03433-7
15. Byappanahalli MN, Nevers MB, Korajkic A, et al. Enterococci in the environment. Microbiol Mol Biol Rev 2012;76(4):685–706. Available from: https://doi.org/10.1128/MMBR.00023-12
16. Gaca AO, Lemos JA. Adaptation to adversity: the intermingling of stress tolerance and pathogenesis in enterococci. Microbiol Mol Biol Rev 2019;83(3):10.1128/mmbr. 00008-19. Available from: https://doi.org/10.1128/MMBR.00008-19.
17. Aktan Y, Tan S, Icgen B. Characterization of lead-resistant river isolate Enterococcus faecalis and assessment of its multiple metal and antibiotic resistance. Environ Monit Assess 2013;185(6):5285–5293. Available from: https://doi.org/10.1007/s10661-012-2945-x.
18. Silva VL da, Caçador NC, Silva C dos SF da, et al. Occurrence of multidrug-resistant and toxic-metal tolerant enterococci in fresh feces from urban pigeons in Brazil. Microbes Environ 2012;27(2):179–185. Available from: https://doi.org/10.1264/jsme2.ME11296
19. De Niederhaeusern S, Bondi M, Anacarso I, et al. Antibiotics and heavy metals resistance and other biological characters in enterococci isolated from surface water of Monte Cotugno Lake (Italy). J Environ Sci Health Part A 2013;48(8):939–946. Available from: https://doi.org/10.1080/10934529.2013.762739
20. Novais C, Campos J, Freitas AR, et al. Water supply and feed as sources of antimicrobial-resistant Enterococcus spp. in aquacultures of rainbow trout (Oncorhyncus mykiss), Portugal. Sci Total Environ 2018;625:1102–1112. Available from: https://doi.org/10.1016/j.scitotenv.2017.12.265
21. Rebelo A, Mourão J, Freitas AR, et al. Diversity of metal and antibiotic resistance genes in Enterococcus spp. from the last century reflects multiple pollution and genetic exchange among phyla from overlapping ecosystems. Sci Total Environ 2021;787:147548. Available from: https://doi.org/10.1016/j.scitotenv.2021.147548
22. Lebreton F, Willems RJ, Gilmore MS. Enterococcus diversity, origins in nature, and gut colonization. In: Gilmore MS, Clewell DB, Ike Y, Shankar N, editors. Enterococci: From Commensals to Leading Causes of Drug Resistant Infection [Internet]. Boston: Massachusetts Eye and Ear Infirmary; 2014–. PMID: 24649513.Available from: https://www.ncbi.nlm.nih.gov/books/NBK190427/
23. Santestevan NA, Angelis Zvoboda D de, Prichula J, et al. Antimicrobial resistance and virulence factor gene profiles of Enterococcus spp. isolates from wild Arctocephalus australis (South American fur seal) and Arctocephalus tropicalis (Subantarctic fur seal). World J Microbiol Biotechnol 2015;31(12):1935–1946. Available from: https://doi.org/10.1007/s11274-015-1938-7
24. Prichula J, Pereira RI, Wachholz GR, et al. Resistance to antimicrobial agents among enterococci isolated from fecal samples of wild marine species in the southern coast of Brazil. Mar Pollut Bull 2016;105(1):51–57. Available from: https://doi.org/10.1016/j.marpolbul.2016.02.071
25. Grassotti TT, Zvoboda DA, Costa LFX, et al. Antimicrobial resistance profiles in Enterococcus spp. isolates from fecal samples of wild and captive black Capuchin Monkeys (Sapajus nigritus) in South Brazil. Front Microbiol 2018;9:2366. Available from: https://doi.org/10.3389/fmicb.2018.02366
26. Huff R, Pereira RI, Pissetti C, et al. Antimicrobial resistance and genetic relationships of enterococci from siblings and non-siblings Heliconius erato phyllis caterpillars. PeerJ 2020;8:e8647. Available from: https://doi.org/10.7717/peerj.8647
27. de Araujo GO, Huff R, Favarini MO, et al. Multidrug resistance in enterococci isolated from wild Pampas Foxes (Lycalopex gymnocercus) and Geoffroy’s Cats (Leopardus geoffroyi) in the Brazilian Pampa biome. Front Vet Sci 2020;7.Available from: https://doi.org/10.3389/fvets.2020.606377
28. Heck JMS, Prichula J, Huff R, et al. Captive snakes from Brazil as carriers of multidrug-resitant enterococci. Int J Plant Anim Environ Sci 2021;11(3):503–523. Available from: https://doi.org/10.26502/ijpaes.202118
29. Schwartzman JA, Lebreton F, Salamzade R, et al. Global diversity of enterococci and description of 18 previously unknown species. Proc Natl Acad Sci 2024;121(10):e2310852121. Available from: https://doi.org/10.1073/pnas.2310852121
30. Ture M, Altinok I, Alp H. Effects of cage farming on antimicrobial and heavy metal resistance of Escherichia coli, Enterococcus faecium, and Lactococcus garvieae. Microb Drug Resist 2018;24(9):1422–1430. Available from: https://doi.org/10.1089/mdr.2018.0040
31. Vignaroli C, Pasquaroli S, Citterio B, et al. Antibiotic and heavy metal resistance in enterococci from coastal marine sediment. Environ Pollut 2018;237:406–413. Available from: https://doi.org/10.1016/j.envpol.2018.02.073
32. Prichula J, Van Tyne D, Schwartzman J, et al. Enterococci from wild Magellanic penguins (Spheniscus magellanicus) as an indicator of marine ecosystem health and human impact. Appl Environ Microbiol 2020;86(19). Available from: https://doi.org/10.1128/AEM.01662-20
33. Mocellin RR, Toigo AL, Santos CC dos, et al. Metal tolerance in enterococci isolated from seabirds in Abrolhos Archipelago, Brazil: Evaluating their role as bioindicators of marine pollution. Mar Pollut Bull 2024;207:116866. Available from: https://doi.org/10.1016/j.marpolbul.2024.116866
34. Instituto Brasileiro De Geografia e Estatística (IBGE). Censo demográfico 2022: Candiota, Rio Grande do Sul. Rio de Janeiro: IBGE, 2022. Available from: https://www.ibge.gov.br/cidades-e-estados/rs/candiota.html. Acesso em: 18 fev. 2026.
35. Prefeitura Municipal de Candiota. A economia local de Candiota. 2023; Available from: https://www.candiota.rs.gov.br/
36. Sikes RS, Gannon WL, Animal care and use committee of the american society of mammalogists. Guidelines of the American Society of Mammalogists for the use of wild mammals in research. J Mammal 2011;92(1):235–253. Available from: https://doi.org/10.1644/10-MAMM-F-355.1
37. Sauget M, Valot B, Bertrand X, et al.Can MALDI-TOF mass spectrometry reasonably type bacteria? Trends Microbiol 2017;25(6):447–455. Available from: https://doi.org/10.1016/j.tim.2016.12.006
38. Cartwright CP, Stock F, Fahle GA, et al.. Comparison of pigment production and motility tests with PCR for reliable identification of intrinsically vancomycin-resistant enterococci. J Clin Microbiol 1995;33(7):1931–1933. Available from: https://doi.org/10.1128/jcm.33.7.1931-1933.1995.
39. Kirby WM, Bauer AW, Sherris JC, et al. Antibiotic susceptibility testing by a standardized single disk method. Am J Clin Pathol 1966;45(4):493–6.
40. Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing. 31st ed. Supplement M100. Wayne, PA: Clinical and Laboratory Standards Institute; 2021.
41. Magiorakos AP, Srinivasan A, Carey RB, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect 2012;18(3):268–281. Available from: https://doi.org/10.1111/j.1469-0691.2011.03570.x
42. Bell JM, Paton JC, Turnidg EJ. Emergence of vancomycin-resistant Enterococcus in Australia: phenotypic and genotypic characteristics of isolates. Journal of Clinical Microbiology, v.36, p.2187-2190, 1998. J. Clin. Microbiol. 1998;36:2187-2190. Available from: https://doi.org/10.1128/jcm.36.8.2187-2190.1998
43. Hasman H, Kempf I, Chidaine B, et al. Copper resistance in Enterococcus faecium, mediated by the tcrB gene, is selected by supplementation of pig feed with copper sulfate. Appl Environ Microbiol 2006;72(9):5784–5789. Available from: https://doi.org/10.1128/AEM.02979-05
44. Sutcliffe J, Grebe T, Tait-Kamradt A, et al. Detection of erythromycin-resistant determinants by PCR. Antimicrob Agents Chemother 1996;40(11):2562–2566. Available from: https://doi.org/10.1128/AAC.40.11.2562
45. Werner G, Hildebrandt B, Witte W. The newly described msrC gene is not equally distributed among all isolates of Enterococcus faecium. Antimicrob Agents Chemother 2001;45(12):3672–3673. Available from: https://doi.org/10.1128/AAC.45.12.3672-3673.2001
46. Frazzon APG, Gama BA, Hermes V, et al. Prevalence of antimicrobial resistance and molecular characterization of tetracycline resistance mediated by tet (M) and tet (L) genes in Enterococcus spp. isolated from food in Southern Brazil. World J Microbiol Biotechnol 2010;26(2):365–370. Available from: https://doi.org/10.1007/s11274-009-0160-x
47. Aarestrup FM, Agerso Y, Gerner–Smidt P, et al. Comparison of antimicrobial resistance phenotypes and resistance genes in Enterococcus faecalis and Enterococcus faecium from humans in the community, broilers, and pigs in Denmark. Diagn Microbiol Infect Dis 2000;37(2):127–137. Available from: https://doi.org/10.1016/S0732-8893(00)00130-9
48. Manfredi C, Lucherini M, Canepuccia AD, et al. Geographical variation in the diet of Geoffroy’s cat (Oncifelis geoffroyi) in Pampas grassland of Argentina. J Mammal 2004;85(6):1111–1115. Available from: https://doi.org/10.1644/BWG-133.1
49. Sousa KS, Bager A. Feeding habits of Geoffroy’s cat (Leopardus geoffroyi) in southern Brazil. Mamm Biol 2008;73(4):303–308. Available from: https://doi.org/10.1016/j.mambio.2007.04.001
50. Trigo TC, Tirelli FP, Machado LF, et al. Geographic distribution and food habits of Leopardus tigrinus and L. geoffroyi (Carnivora, Felidae) at their geographic contact zone in southern Brazil. Stud Neotropical Fauna Environ 2013;48(1):56–67. Available from: https://doi.org/10.1080/01650521.2013.774789
51. Migliorini RP, Peters FB, Favarini MO, et al. Trophic ecology of sympatric small cats in the Brazilian Pampa. PLoS One 2018;13(7):e0201257. Available from: https://doi.org/10.1371/journal.pone.0201257
52. Pereira JA, Aprile G. Gato montés (Oncifelis geoffroyi).In: Felinos de Sudamérica: Manuales de campo. Londaiz Laborde Ediciones. Londaiz Laborde Ediciones. Buenos Aires; 2012. Cap. VII. p. 64-69. Espanhol.
53. Lauková A, Strompfová V, Simonová M, et al. Species of Enterococcus faecalis associated with free-living rodents. Folia Microbiol (Praha) 2008;53(2):173–178.
54. Poeta P, Costa D, Sáenz Y, et al. Characterization of antibiotic resistance genes and virulence factors in faecal enterococci of wild animals in Portugal. J Vet Med 2005;52(9):396–402. Available from: https://doi.org/10.1111/j.1439-0450.2005.00881.x
55. Cassenego APV, Ellwanger J, Azevedo PA d’, et al. Virulence and biofilm formation by Enterococcus faecalis isolates from cloacal swabs of broilers infected with Eimeria spp. Pesq Vet Bras 2013;33:1433–1440. Available from: https://doi.org/10.1590/S0100-736X2013001200007
56. Silva L da, Grecellé CZ, Frazzon APG, et al. Multidrug-resistant Enterococcus faecium and Enterococcus faecalis isolated from dogs and cats in southern Brazil. Microbiol Res 2024;15(3):1083–1090. Available from: https://doi.org/10.3390/microbiolres15030071
57. Grenni P, Ancona V, Caracciolo AB. Ecological effects of antibiotics on natural ecosystems: A review. Microchem J 2018;136:25–39. Available from: https://doi.org/10.1016/j.microc.2017.02.006
58. Santana CB dos S, Mattos MCC de, Paz RS, et al. Análise do consumo de cloridrato de tetraciclina para uso veterinário no Brasil. Rev Contemp 2023;3(12):26378–26395. Available from: https://doi.org/10.56083/RCV3N12-086
59. Portella DN, Faria EC, Hennequin IS, et al. Impactos causados no meio ambiente por resíduos antimicrobianos de uso veterinário: Impacts caused on the environment use antimicrobial waste. Braz J Anim Environ Res 2023;6(4):3708–3722. Available from: https://doi.org/10.34188/bjaerv6n4-049
60. Silva RA da, Luiza VL, Bermudez JAZ, et al. The One Health approach to face bacterial resistance to antibiotics in livestock production. Saúde Em Debate 2025;49:e9713. Available from: https://doi.org/10.1590/2358-289820251449713i
61. Clewell DB, Flannagan SE, Jaworski DD. Unconstrained bacterial promiscuity: the Tn916–Tn1545 family of conjugative transposons. Trends Microbiol 1995;3(6):229–236. Available from: https://doi.org/10.1016/S0966-842X(00)88930-1
62. Chopra I, Roberts M. Tetracycline antibiotics: mode of action, applications, molecular biology, and epidemiology of bacterial resistance. Microbiol Mol Biol Rev 2001;65(2):232–260.Available from: https://doi.org/10.1128/MMBR.65.2.232-260.2001
63. Souza JM, Carneiro MF, Paulelli ACC, et al. Arsenic and rice: toxicity, metabolism, and food safety. Quím Nova 2015; 38(1): 118-127. Available from: https://doi.org/10.5935/0100-4042.20140279
Downloads
Arquivos adicionais
Publicado
Como Citar
Edição
Seção
Licença
Copyright (c) 2026 Ciência Animal Brasileira / Brazilian Animal Science

Este trabalho está licenciado sob uma licença Creative Commons Attribution 4.0 International License.
Autores que publicam nesta revista concordam com os seguintes termos:
- Autores mantém os direitos autorais e concedem à revista o direito de primeira publicação, com o trabalho simultaneamente licenciado sob a Licença Creative Commons Attribution que permite o compartilhamento do trabalho com reconhecimento da autoria e publicação inicial nesta revista.
- Autores têm autorização para assumir contratos adicionais separadamente, para distribuição não-exclusiva da versão do trabalho publicada nesta revista (ex.: publicar em repositório institucional ou como capítulo de livro), com reconhecimento de autoria e publicação inicial nesta revista.
- Autores têm permissão e são estimulados a publicar e distribuir seu trabalho online (ex.: em repositórios institucionais ou na sua página pessoal) a qualquer ponto antes ou durante o processo editorial, já que isso pode gerar alterações produtivas, bem como aumentar o impacto e a citação do trabalho publicado (Veja O Efeito do Acesso Livre).
Declaração de dados
-
Os dados de pesquisa estão contidos no próprio manuscrito


























